Factory
In the industry's most advanced clean room, strict quality control throughout the manufacturing process
- ○ A factory where the flow of people, goods, and air is thoroughly controlled
- ○ System design to ensure and maintain high quality
- ○ Well-organized responsibility system for quality control minimizes human error in manufacturing
- ○ Prevention of contamination and deterioration of quality
We firmly control the flow lines of people, goods, and air, and deter wasteful movement to prevent quality deterioration. In addition to sanitary materials, we have state-of-the-art facilities that can clear strict quality and safety standards for pharmaceuticals and medical devices. In the future, there will be a growing demand for highly specialized products in a wide range of fields at medical and nursing care sites. Therefore, we respond to variations in size, folding method, and packaging, thoroughly manage the product, and perform integrated production from the arrival of raw materials to processing and storage.
Factory features

"Positive pressure" prevents outside air from entering and maintains a clean-air manufacturing environment
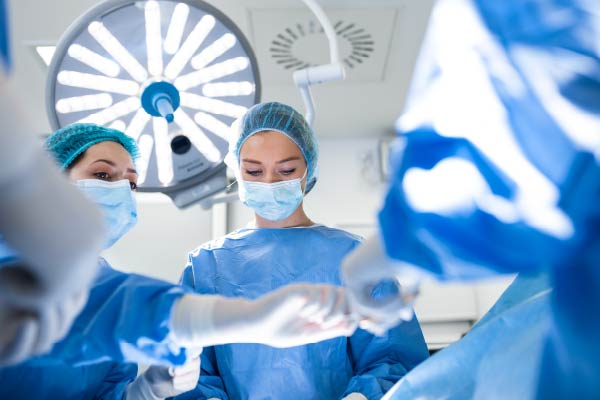
"Positive pressure" means that the inside air pressure is higher than the outside air. Air pressure has the characteristic of flowing from high pressure to low. Increasing the amount of air supplied to the room and making the interior air pressure higher than the outside air to maintain clean air is called "positive pressure". This prevents the inflow of foreign matter from the outside. This technology, which is also used in hospital operating rooms and ICUs, supports the excellent hygiene management capabilities of Oguchi Eizai's clean rooms.
Business
Medical
We fold non-woven gauze for medical use, special gauze used in dialysis kits, and wipes for medical instruments.
Cosmetics

We manufacture high-quality cotton puffs used by makeup artists, hair salons, beauty salons, etc.
Industrial

We develop and process wipers for industrial precision work in a clean environment
























